All articles by Manish Kumar
Blood test may detect stroke type before hospital arrival, study finds
Researchers at the American Heart Association tested glial fibrillary acidic protein blood levels to quickly diagnose stroke types, potentially reducing delays in treatment and brain damage
Breakthrough technology for human muscle fatigue measure, Myocene, gets FDA “de novo” approval
MYOCENE kicks off the first FDA De Novo clearance of 2025 for its groundbreaking muscle fatigue monitoring device.
Stereotaxis announces first GenesisX Robotic System order
GenesisX is the latest advance in endovascular surgical robotics, building upon the established benefits of Robotic Magnetic Navigation while significantly enhancing the accessibility of the technology for healthcare providers
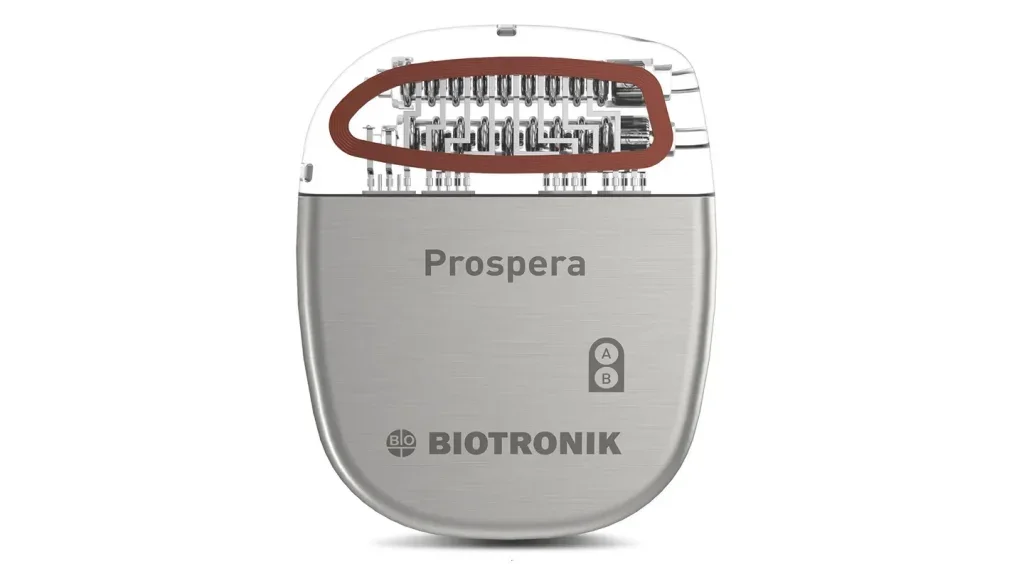
Biotronik’s Prospera SCS system shows pain relief potential in BENEFIT-03 trial
The FDA-approved Prospera SCS system optimises chronic pain management with RESONANCE stimulation paradigm, remote monitoring, and proactive care
Aethlon Medical begins Australian Hemopurifier trial with first patient treatment
The Aethlon Hemopurifier is an experimental extracorporeal device aimed at filtering enveloped viruses and tumour-derived extracellular vesicles from the bloodstream
Life Spine announces FDA 510(k) clearance for the ProLift Pivot Expandable Spacer System
The ProLift Pivot System represents the latest innovation in Life Spine’s Micro Invasive technology portfolio
HeartBeam achieves major milestone with FDA submission for its 12-lead ECG synthesis software
The FDA submission is backed by robust data from the VALID-ECG pivotal study, which enrolled 198 patients across five clinical sites
Stryker to divest US spinal implants division to Viscogliosi Brothers
The agreement includes a binding offer to acquire Stryker’s spinal implants business in France and create a new company called VB Spine
GE HealthCare gets FDA nod for updated Voluson Expert ultrasound systems
The Voluson Expert 22, 20, and 18 offer high-resolution imaging, advanced technology, and an ergonomic design to enhance workflow efficiency and user experience
Beckman Coulter receives FDA breakthrough device designation for Alzheimer’s disease blood test
This blood test is designed to aid healthcare providers identify patients with amyloid pathology associated with Alzheimer’s disease