White Papers
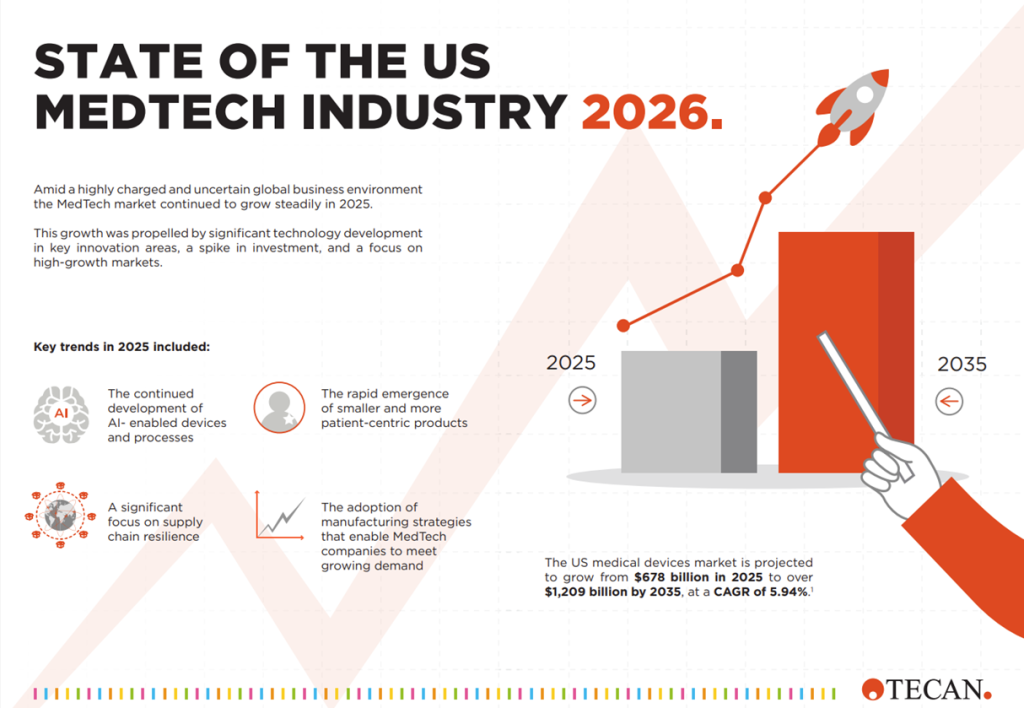
State of the US Medtech Industry 2026
Amid a highly charged and uncertain global business environment the MedTech market continued to grow steadily in 2025. This growth …
No room for error
Designed for zero-defect mechanical assembly, Tecan’s vPoke® technology brings the highest quality healthcare innovations to life.
Transferring Complex Builds into Reliable Production
First to market competition. Navigating geopolitical disruptions. Integrating rapidly emerging technologies into next-gen products. Tecan’s seamless bridge from design to …
Understanding the Regulatory Difference Between SiMD and SaMD
As digital technology continues to reshape healthcare, the line between hardware and software in medical devices has blurred. From embedded …
The Importance of Measuring Intraocular Pressure Correctly
Like most complex systems, the human eye occasionally malfunctions with potentially grave effects on lifestyle and mental health[1]. To assess …
Delivering the most viscous drug ever put in an autoinjector
Strategic and technical help with the autoinjector design
Does My Medical Device Design Actually Work?
There are few experiences that can surpass the joy of seeing a product you developed reach manufacture.
The Rise of the Next-Gen CDMO: Strategies for Reducing Risk and Time to Market
Creating and producing advanced drug-device combinations, like autoinjectors and pen injectors, requires a depth and breadth of technical expertise. As …
Medical Sensor Technology
How Electromechanical and Fiber Optic Sensors can be used to collect data in Medical Devices. Receive Better Feedback, Improve Patient …
Fiber Optic Sensors for Medical Devices
As medical procedures continue to advance with the development of new technologies and manufacturing capabilities, MedTech and life sciences engineers face …
Nitinol Post‑Shape‑Setting Time Temperature Transformation (TTT) and Time Temperature Stress (TTS) Properties
Abstract The tuning of Nitinol components to achieve target transformation temperatures and mechanical properties requires careful selection of shape-setting recipes. …
Introduction to Nitinol
This whitepaper offers a clear and informative introduction to nitinol and its growing impact on the medical device industry. It …
MICROFLUIDICS: Laminate tolerancing, capability and measurements in laser processing
Medical designers engaged in the creation of Point-of-Care microfluidic test devices have a strong interest in knowing the capability of …
Sealed for success
Medical Device Developments sits down with Charlotte Mavor, strategic programme leader at Oliver Healthcare Packaging, to discuss peelable sealants, their …