All articles by Manish Kumar
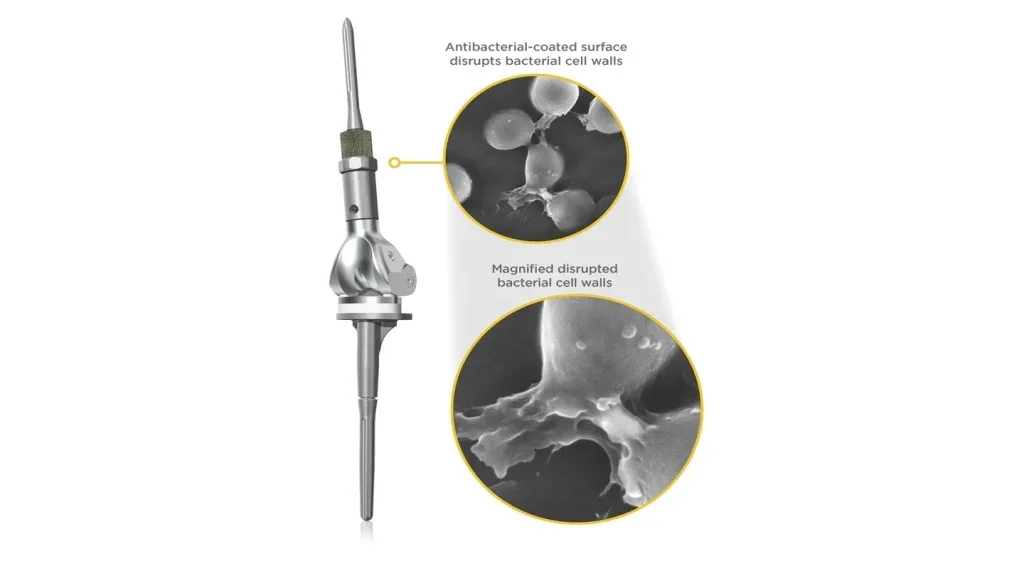
Onkos Surgical successfully completes groundbreaking, first-in-human cases using NanoCept antibacterial technology
NanoCept is a novel antibacterial coating applied to orthopaedic implants to help address intraoperative bacterial contamination
SynCardia Systems receives decision to grant first patent in China for the emperor next-generation total artificial heart
This patent describes novel technology aimed at eliminating external drive mechanisms by fully integrating the pumping mechanism within the implanted artificial heart ventricle
KARL STORZ acquires T1V’s medical business to enhance digital surgery
The acquisition will enhance KARL STORZ’s digital surgery solutions, integrating T1V’s expertise, patents, and technologies into its expanding ecosystem over the next year
AerWave completes FIH study for ultrasound lung denervation in COPD and asthma
The US-based medical device company has also announced the raising of $12m in funding, led by Lucius Partners
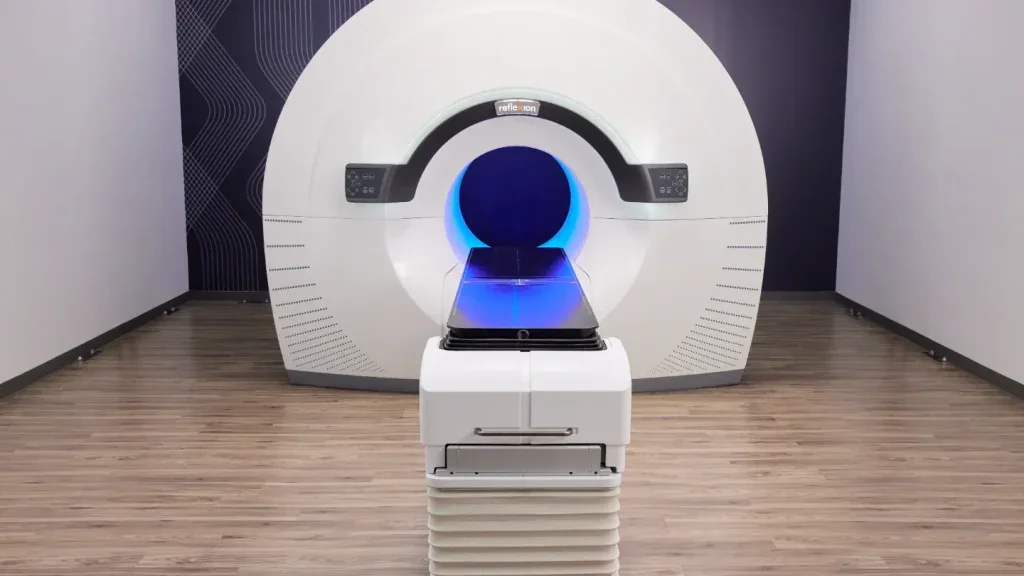
RefleXion launches pivotal study to expand SCINTIX therapy and announces broader medicare reimbursement
Hackensack Meridian John Theurer Cancer Center initiates new clinical study
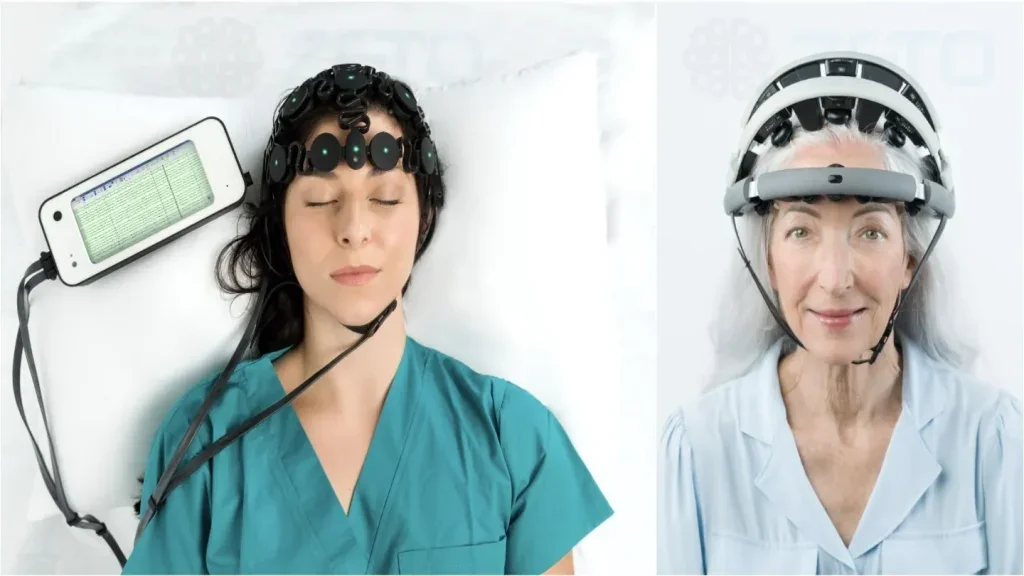
Zeto secures $31m in Series B funding to redefine the future of EEG brain monitoring
The funding round was led by MindWorks Global (MWG), a Michigan-based investment entity funded by visionary entrepreneur-investors with a track record of building multi-billion-dollar enterprises
Pacira gets FDA clearance for new iovera° Smart Tip to manage back pain
ioveraº is a drug-free, handheld device delivering controlled cold therapy to target nerves, providing immediate, long-lasting relief for back pain by accessing medial branch nerves
Anumana partners with AliveCor to enhance cardiac detection with AI
Both partners will collaborate to integrate AI algorithms for early cardiac disease detection, starting with the FDA-cleared ECG-AI LEF algorithm on Kardia ECG devices
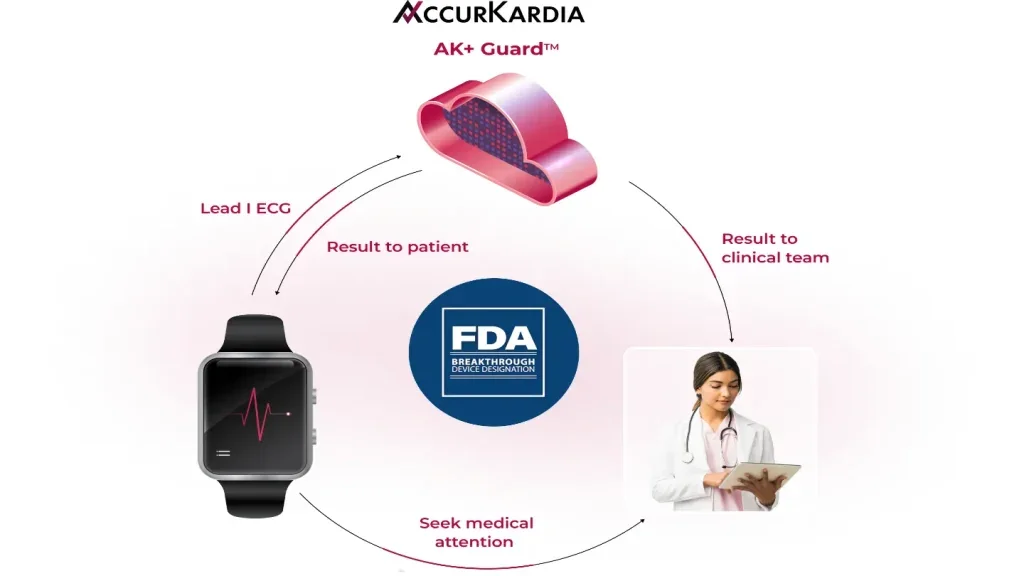
AccurKardia receives FDA breakthrough device designation for AI-powered hyperkalemia detection solution using lead I ECG data
AK+ Guard also accepted into the FDA Total Product Life Cycle Advisory Program
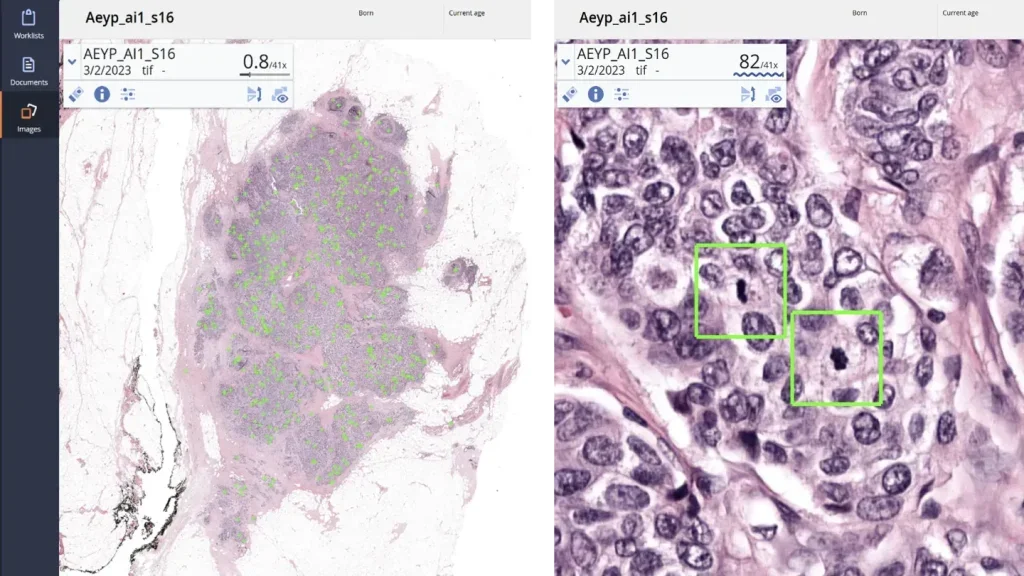
Aiosyn Mitosis Breast becomes the first AI-powered mitosis detection solution to achieve CE mark certification under IVDR
This milestone establishes Aiosyn Mitosis Breast as the first IVDR-certified application for mitosis counting in breast cancer diagnostics and accelerates the company’s growth in clinical pathology