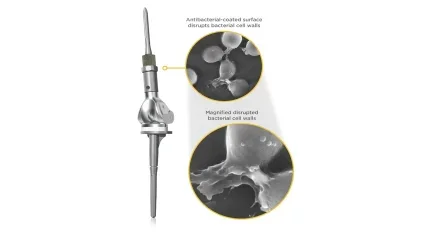
Onkos Surgical, a leading provider of innovative solutions for complex orthopaedic procedures, today announced that its revolutionary NanoCept Antibacterial Technology has been used successfully in multiple surgical procedures. NanoCept is a novel antibacterial coating applied to orthopaedic implants to help address intraoperative bacterial contamination.
“Limb salvage and revision procedures are challenging, and the rate of complication is much higher than most index orthopaedic procedures,” said Steven Gitelis, MD, Director of Musculoskeletal Oncology at Rush University Medical Center and Chief Medical Officer of Onkos Surgical. “The risk of implant infection is on my mind every time I take a patient to the operating room, and this is especially true for patients with bone cancer, as they are more susceptible. The NanoCept Technology represents a significant advancement in the preventative measures my colleagues and I use to mitigate the potentially devastating complications related to intraoperative bacterial contamination of implants. It has the potential to create a paradigm shift in implant selection and is a valuable tool surgeons will now have that may help address an unsolved clinical problem.”
Orthopaedic implant-related infections are clinically challenging and have significant negative effects on patients. Periprosthetic joint infection (PJI) has a lower survival rate than several forms of cancer and is expected to cost the U.S. healthcare system up to $1.8 billion dollars by 2030. It is estimated that 60-70% of all early and delayed-onset PJI (<3 months and 12-24 months post-surgery) are initiated at the time of the procedure through intraoperative contamination.
Onkos received De Novo market authorization from the Food and Drug Administration (FDA) in April of 2024 for its NanoCept Antibacterial Technology. Data submitted to support the De Novo application showed that NanoCept has up to a 99.999% (5-log reduction) kill rate in standardized in-vitro testing of bacteria that are commonly found in the operating room environment. The initial FDA authorization for NanoCept was for the Company’s ELEOS Limb Salvage System, which is a modular total joint system used for patients with bone loss.
“The successful completion of these initial surgeries is a key achievement and significant milestone in our strategy to deploy the NanoCept Technology across our portfolio,” said Patrick Treacy, Onkos CEO and Co-founder. “We believe NanoCept is a platform technology that will substantially extend our reach and capability in broad orthopaedic markets, and we are actively working to expand regulatory clearances beyond ELEOS to include implants used in orthopaedic reconstruction due to tumor, trauma and other orthopaedic revision procedures.”






