All articles by Sdebbarma Sdebbarma
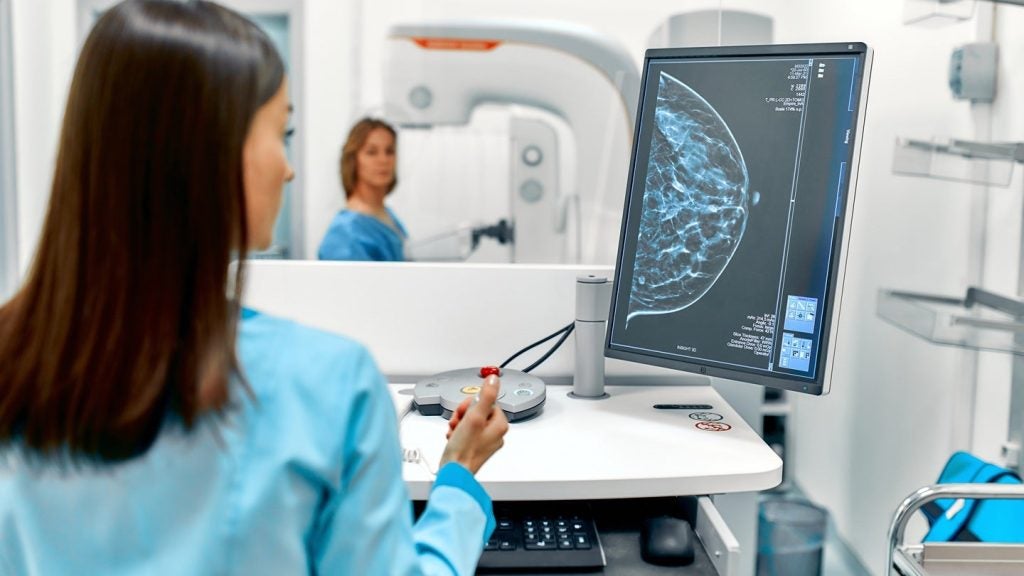
GE and DeepHealth expand partnership for breast screening solutions
The agreement builds upon a 2024 collaboration integrating DeepHealth’s AI workflow with GE HealthCare’s Senographe system.
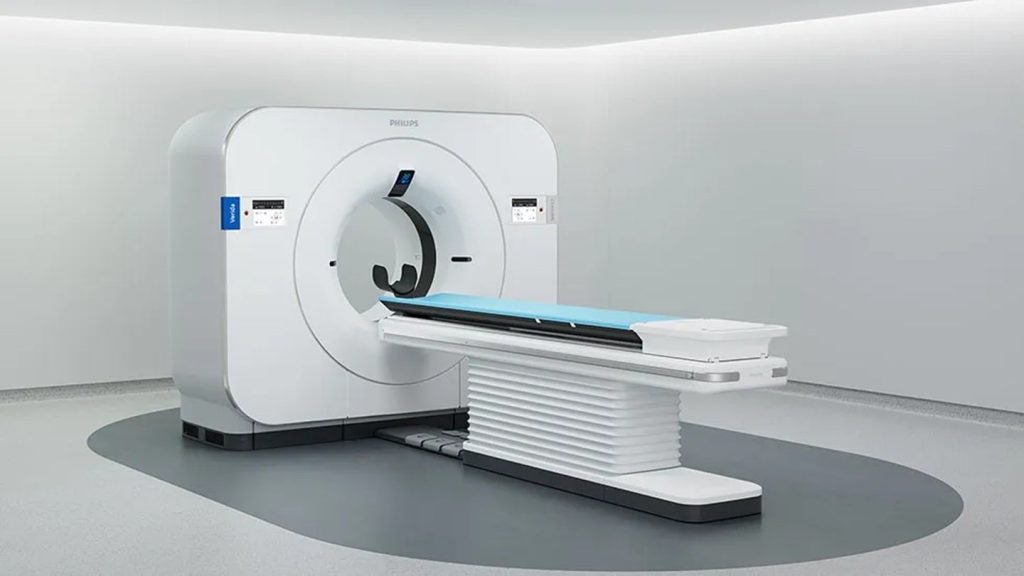
Philips secures FDA clearance for Verida spectral CT system
Verida enables uninterrupted spectral imaging, removing the need for separate scans or workflow changes.
Stereotaxis to acquire Robocath for up to $45m
The acquisition is intended to strengthen Stereotaxis’ position in the sector by expanding its platform.
FDA grants Protaryx Medical clearance for transseptal puncture device
The system offers zero-exchange delivery with a unique atraumatic design to reduce steps and eliminate rewiring during access.
AIP to acquire Avanos Medical for $1.27bn
After closing, Avanos will be private in Alpharetta, Georgia, and delist its shares from the NYSE.
ProSomnus secures FDA clearance for RPMO2 device
RPMO₂ features a fully embedded oximeter over maxillary dentition, using buccal vestibule mucosa behind the upper lip.
J&J reports 12-month data for Varipulse PFA platform
The complete study cohort includes 1,023 patients from 22 sites in Europe.
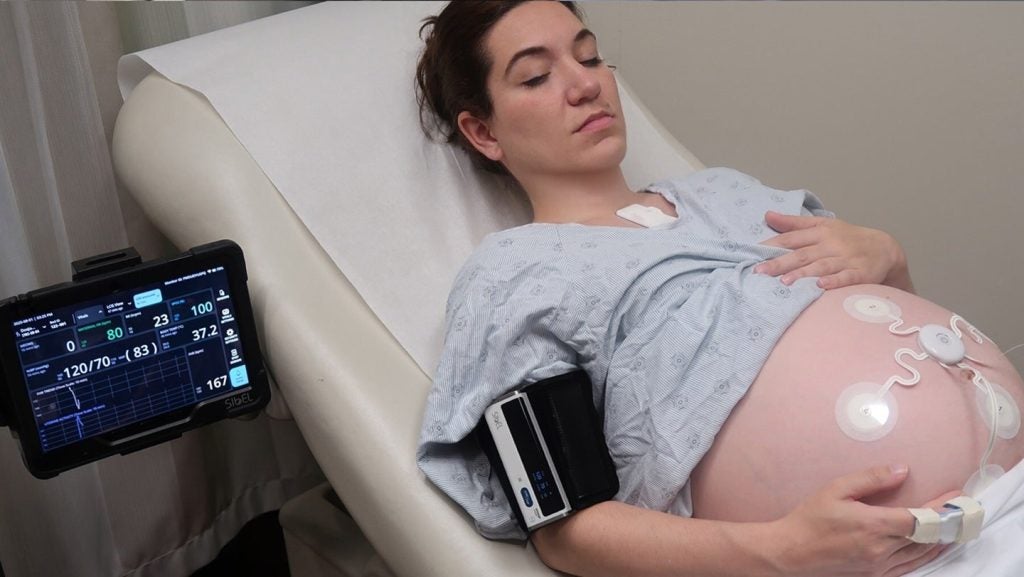
FDA clears Sibel Health’s ANNE Maternal wireless system
ANNE Maternal is a sister platform to ANNE One, which has also been cleared by the FDA for adolescent and adult monitoring.
GE HealthCare and Medtronic integrate bkActiv iUS and Stealth AXiS system
Through the integration, neurosurgeons gain a “plug-and-play” real-time visualisation tool using bkActiv and Stealth AXiS.
MiRus treats first patients in STAR trial of Siegel 8-French heart valve
The randomised controlled, multi-centre, prospective trial will enrol 1,025 patients across multiple centres in the US.