All articles by Manish Kumar
Murata’s revolutionary stretchable printed circuit elevates wearable medical devices
It is perfect for creating advanced medical products, like wearable therapeutic devices and vital monitoring tools, that can provide better accuracy, durability, and patient comfort than today’s equivalent devices
LumiThera obtains FDA authorisation of Valeda Treatment for dry AMD patients to improve vision
The Valeda therapy is the first ever authorized treatment by the FDA for vision loss in dry AMD patients
Medeologix and Biocoat expand catheter solutions with hydrophilic coating deal
Through the agreement, Medeologix enhances its one-stop catheter manufacturing solutions by integrating hydrophilic coating services, boosting efficiency, quality, and cost-effectiveness
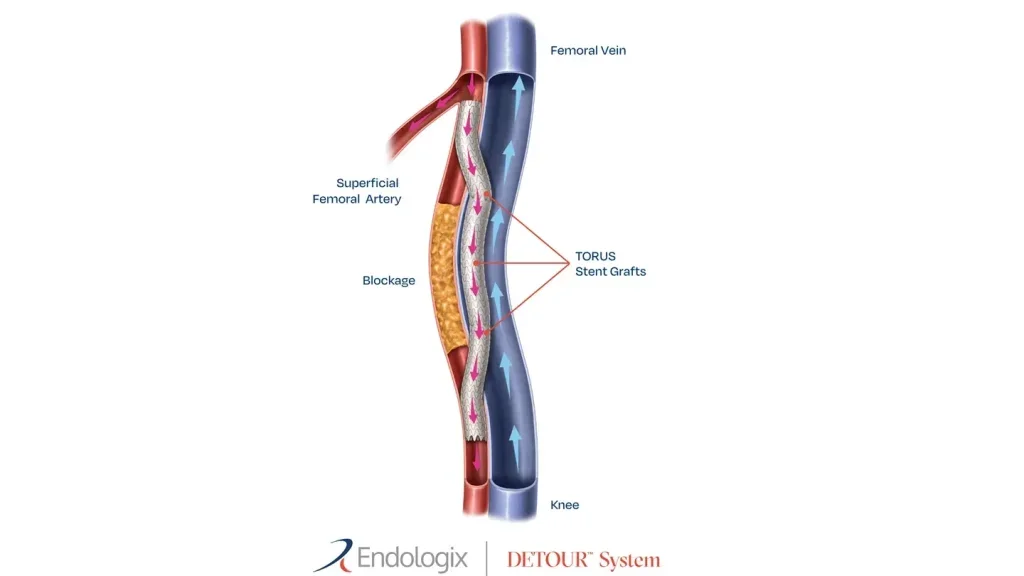
Endologix reports positive long-term results of DETOUR System for PAD
The DETOUR System, featuring ENDOCROSS device and TORUS stent grafts, allows physicians to bypass lesions in the superficial femoral artery using stents to restore blood flow to the leg
Avanos Medical launches new CORGRIP SR
Advancement to reduce dislodgement in feeding tube retention systems
Abbott’s Esprit BTK system shows long-term efficacy in severe PAD treatment
The system demonstrated that 90.3% of patients avoided re-intervention and showed improved outcomes over balloon angioplasty in treating severe peripheral artery disease below the knee
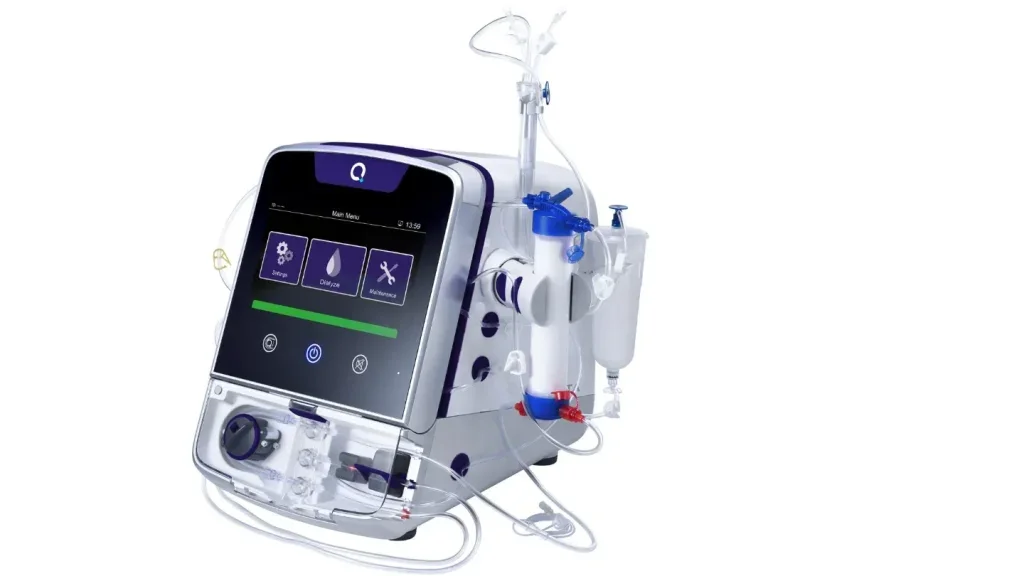
Quanta Dialysis Technologies gets FDA clearance for Quanta Dialysis System
The home dialysis system offers a high-flow dialysis solution for patients with acute or chronic renal failure, with or without ultrafiltration, in both acute and chronic care settings
Medtech developer StarFish Medical acquires California-based Omnica Corporation, strengthening base in a major market
This is the company’s second acquisition in seven years and brings StarFish Medical closer to clients in California — a major center of medical device innovation
NeurAxis receives new FDA 510(K) clearance for IB-Stim, expanding its addressable market
Clearance expands indication for use from 11-18 year olds to 8-21 year olds; and from 3 devices, not to exceed 4 weeks, to 1 device per week for 4 weeks
US Army trials Prytime’s pREBOA-PRO catheter for combat casualty treatment
The study will assess the catheter’s effectiveness in managing severe haemorrhage, and improving care for critically injured patients in challenging combat environments