All articles by Manish Kumar
Health Canada approves Aktiia’s blood pressure monitoring bracelet
The device provides up to 800 automatic blood pressure readings per month, offering insights that can be shared with doctors to track medication and lifestyle impacts on health
Stryker unveils Oculan Lighting Platform to enhance surgical visualisation and illumination
The Oculan Lighting Platform offers advanced features designed to enhance surgical precision and comfort
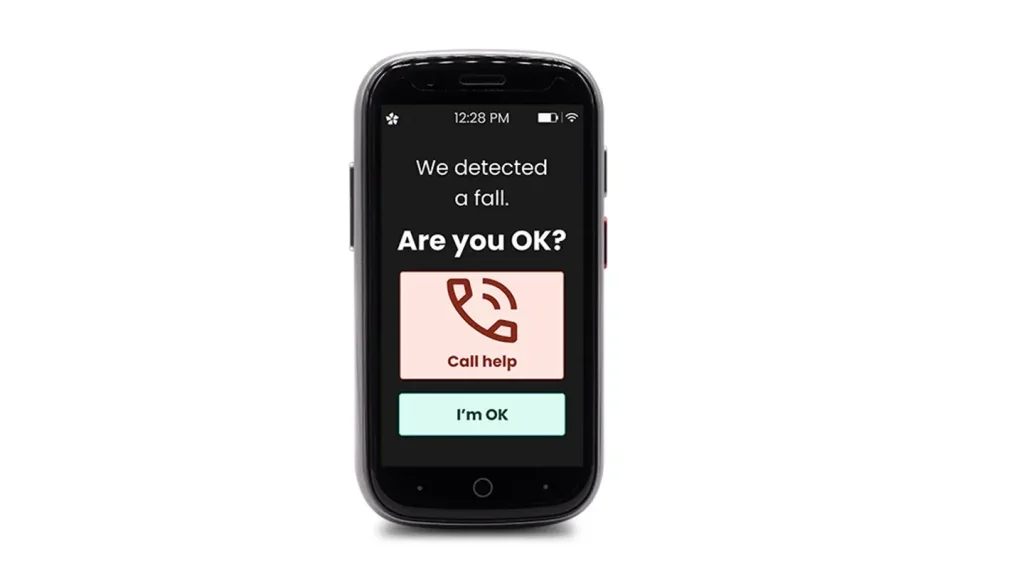
LogicMark launches revolutionary medical alert device, Freedom Alert Max, offering cellular communication technology and 24/7 assistance
The Freedom Alert Max empowers seniors through proprietary, 4G LTE mobile technology as the first fall detection device with emergency video for caregivers, regular over-the-air feature updates, and geofencing for patients with Alzheimer’s and dementia
Roche secures CE mark for VENTANA FOLR1 assay in ovarian cancer treatment
The test identifies eligibility for Elahere, an ADC therapy by AbbVie, which recently received EC marketing authorisation for FRɑ-positive, platinum-resistant ovarian cancer patients
Abbott reports long-term benefits of Amplatzer Amulet in AFib stroke prevention
The Amplatzer Amulet is a heart closure device that prevents blood clots in the left atrial appendage, reducing stroke risk for patients with atrial fibrillation
C Ray Therapeutics completes over $100m Series A+ financing, jointly led by SCGC and TaiLong Capital
C Ray Therapeutics, established in 2021 and headquartered in Chengdu Medical City, is incubated by internationally renowned investment institutions 6 Dimensions Capital
THINK Surgical receives FDA 510(k) clearance for use of LinkSymphoKnee (LSK) implants with TMINI miniature robotic system
The LinkSymphoKnee has been added to THINK Surgical’s ID-HUB, a proprietary data bank of implant modules for use with the open platform version of its TMINI System in the United States
Augurex Life Sciences and Quest Diagnostics partner on RA diagnosis
Quest Diagnostics will offer a 14-3-3η biomarker test, starting 2 December 2024, through its labs and 2,000+ patient service centres across the US, with a physician’s order
Cofactor Genomics launches OncoPrism-NSCLC for lung cancer patients
OncoPrism-NSCLC, validated with data from 12 PREDAPT trial sites, accurately predicts ICI response in NSCLC patients, including monotherapy and chemotherapy combinations
Christ Hospital surgeons perform fourth-in-the-world implant of the BiVACOR total artificial heart
The Christ Hospital is one of only four sites in the nation currently enrolling patients in an FDA-approved clinical trial