All articles by Manish Kumar
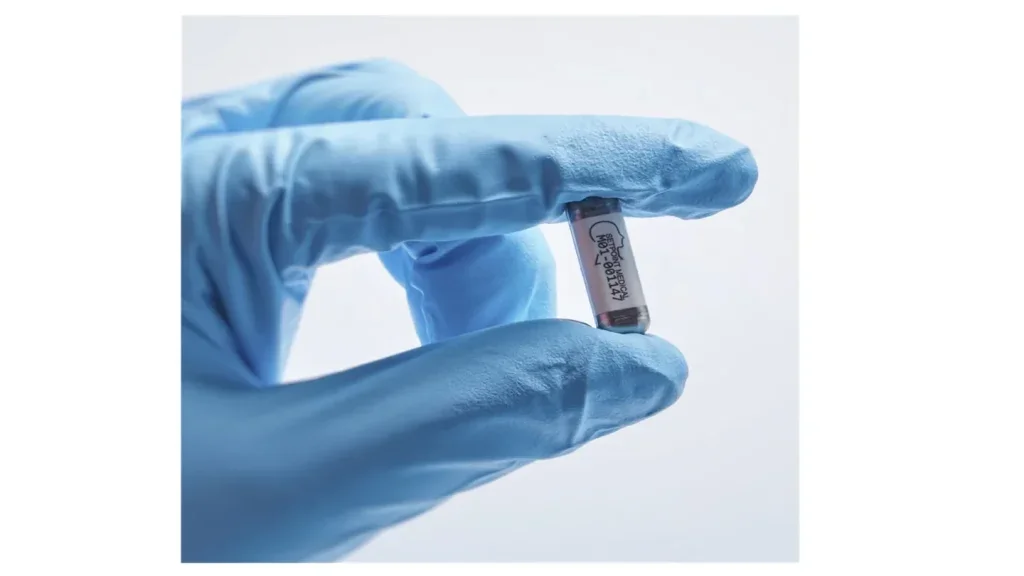
FDA grants breakthrough status to MeMed Severity test for sepsis
MeMed Severity aids clinicians in managing suspected sepsis and acute infections, providing faster, data-driven insights for improved decision-making and patient care
Inspira expands HYLA blood sensor technology into personalised blood lab testing for rapid detection of serious medical conditions
The HYLA technology could also potentially reduce countless tests performed each year where valuable time is often lost between the time of ordering a blood test and when test results are received, which can impact patient treatment and outcomes
Siemens Healthineers acquires Advanced Accelerator Applications Molecular Imaging
Deal extends our network of PET radiopharmaceuticals production sites across Europe
SetPoint Medical files FDA PMA application for SetPoint System in RA treatment
SetPoint System is a neuroimmune modulation device designed to be used in adults with moderate-to-severe RA, who are unresponsive to or intolerant of biologic or targeted synthetic DMARDs
PainTEQ secures new patent for LinQ SI joint stabilisation system
The patent introduces two key advancements in instrument alignment and protects features that improve procedural precision, efficiency, and patient safety
Cala Health closes oversubscribed $50m financing to propel growth
Cala is continuing to build strong clinical evidence to expand patient access for our neurostimulation therapy
Artivion granted FDA Humanitarian Device Exemption for the AMDS hybrid prosthesis
The AMDS is the world’s first aortic arch remodeling device for use in the treatment of acute DeBakey Type I aortic dissections
Adaptive Biotechnologies reports new MRD data for clonoSEQ test in blood cancer
clonoSEQ is an FDA-cleared and CE-marked test for detecting MRD in multiple myeloma, B-ALL, and CLL patients using bone marrow or blood samples
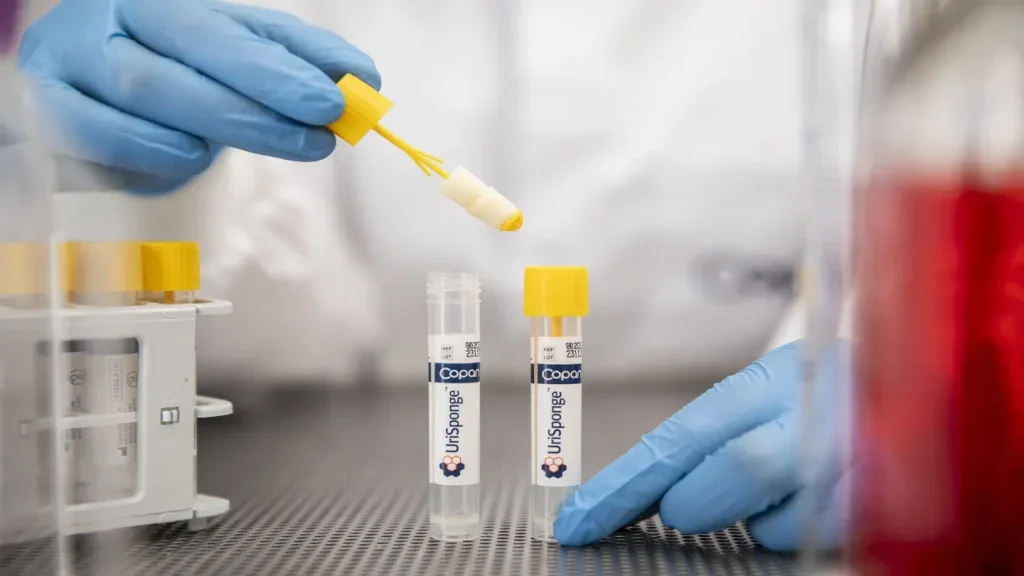
Copan Diagnostics’ UriSponge urine collection device gets FDA clearance
UriSponge is intended to provide a cost-effective, user-friendly solution for improving urine specimen collection, preservation, and transport using advanced preservatives
Sonic Healthcare to acquire LADR Laboratory Group in Germany
LADR is a national laboratory participant, providing highquality testing services delivered through a network of stand-alone and hospital-based laboratories throughout Germany