All articles by Manish Kumar
QHSLab and South Dakota State University celebrate NSF invitation to submit STTR application for AIR-GUIDDE technology
This exclusive invitation, under the NSF Small Business Technology Transfer (STTR) program, underscores the transformative potential of AIR-GUIDDE and highlights the significant merits of the project, even at this early stage
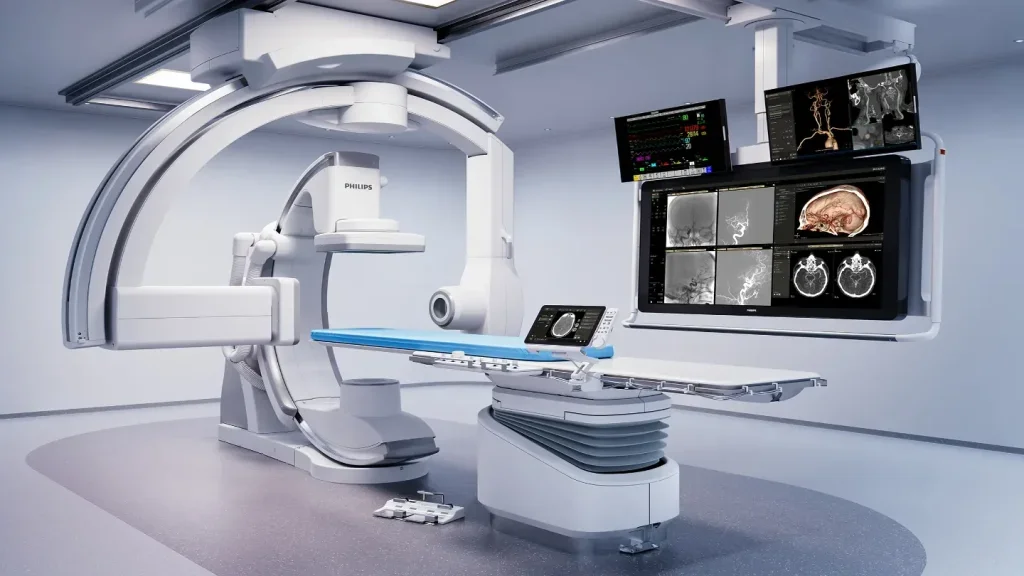
Philips expands deal with Sim&Cure to boost neurovascular solutions
The collaboration will integrate Sim&Size software into Azurion to strengthen Philips’ position in interventional neuro solutions and enhance image-guided therapy capabilities
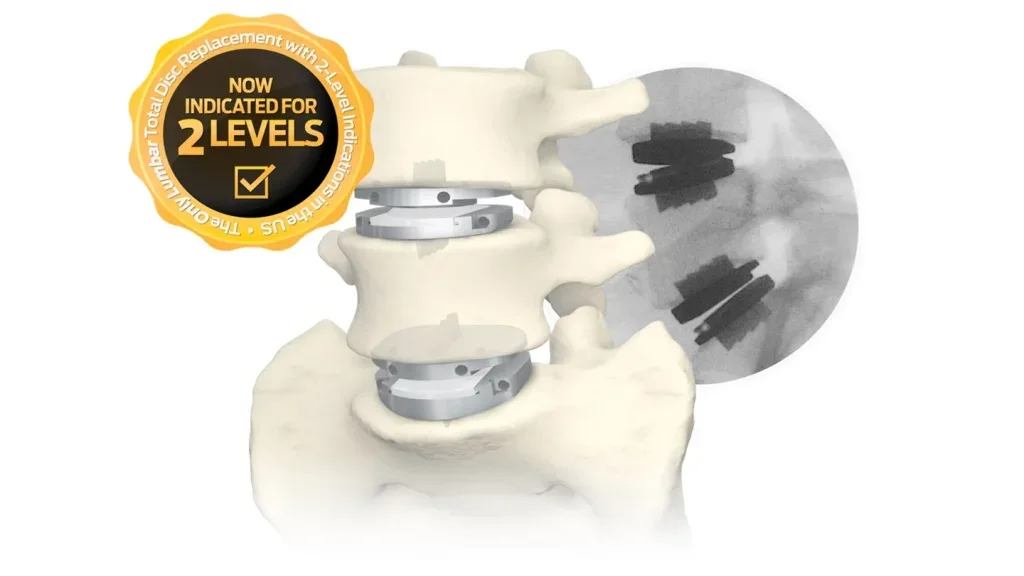
Centinel Spine reports long-term positive results of prodisc L system
The study validates prodisc L’s fixed-core design, showing low revision and adjacent-level surgery rates, with equivalent outcomes for 1- and 2-level LTDR, and prior surgery patients
PanGIA Biotech announces landmark partnership agreement for first liquid biopsy prostate cancer assay in India
Revolutionizing Prostate Cancer Detection with a Simple Urine Test
LuminoDx announces CLIA certification for San Diego laboratory
CLIA certification, issued by the Centers for Medicare and Medicaid Services (CMS), is required for clinical laboratories to accept and test human samples for diagnostic purposes
Capstan Medical raises $110m in Series C for heart valve solutions
The heart valve intervention provider will use the new funding to advance its structural heart treatments and accelerate clinical and development efforts for upcoming trials
Mindset Medical gets FDA clearance for contactless pulse rate IVC app
The IVC App allows the remote collection of pulse rate measurements using patients’ own devices, offering easy access to health data at home, in offices, or healthcare settings
Osteotec appointed as exclusive distributor for Cerapedics in the UK and Ireland
This agreement expands Osteotec’s extensive offering, providing healthcare professionals with access to Cerapedics’ i-FACTOR Bone Graft Putty and i-FACTOR Bone Graft FLEX FR products for use in spine procedures
FemPulse receives FDA IDE approval for the pivotal clinical trial of its wearable bioelectronic device for overactive bladder in women
FemPulse Corporation is a bioelectronic medicine company developing a novel wearable bioelectronic platform for the personalized treatment of medical conditions in women
Agilent wins IVDR certification for PD-L1 IHC 28-8 pharmDx assay
PD-L1 IHC 28-8 pharmDx provides key insights into PD-L1 expression, aiding in predicting response to anti-PD-1 therapies like Opdivo and Opdualag across various cancer types