All articles by Manish Kumar
Alleviant raises $90m for second trial of heart failure atrial shunt
The medical device firm conduct the ALLAY-HFrEF trial, a sham-controlled study of its no-implant atrial shunt for heart failure patients with reduced ejection fraction
Adaptive Biotechnologies, NeoGenomics partner on personalised disease monitoring
The partnership combines Adaptive’s clonoSEQ MRD with NeoGenomics’ COMPASS and CHART services to provide valuable insights throughout the treatment process
icotec announces FDA clearance for spinal infection indication with BlackArmor implants
icotec is honored to be the first and only company in the United States with FDA 510(k) clearance for stabilizing the spine in de novo spinal infections, including discitis, osteomyelitis, pyogenic infection of the intervertebral disc, and other spondylopathies
Haemonetics completes sale of whole blood assets to GVS
The transaction comprises a total cash consideration of up to $67.8M, including $45.3M upfront after giving effect to certain customary adjustments, and up to $22.5M in contingent earn-outs over the next four years
Bayer announces positive Phase 3 results for gadoquatrane MRI agent
Gadoquatrane met all the primary and secondary endpoints in QUANTI studies, showing 60% less gadolinium compared to standard macrocyclic GBCAs
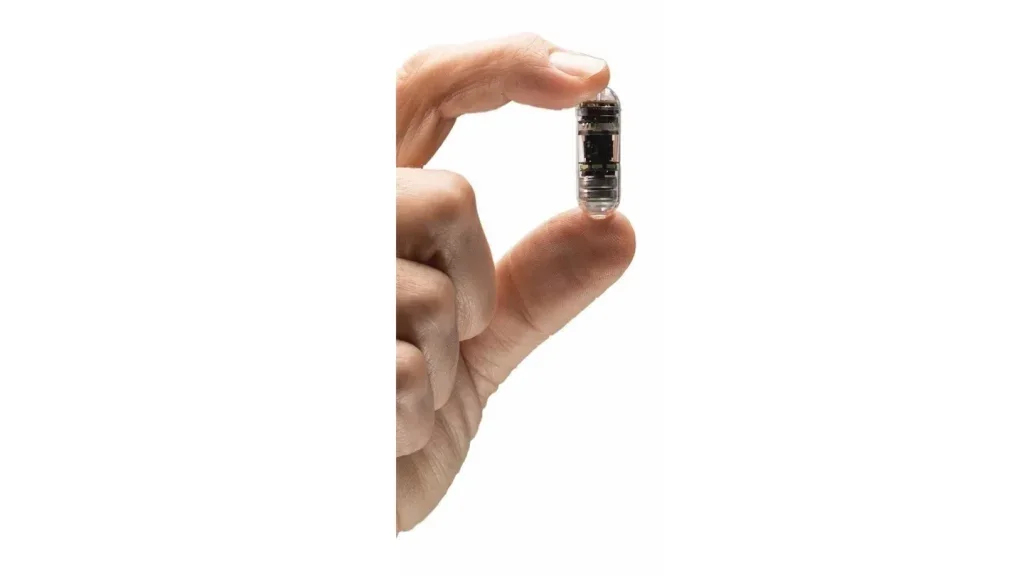
CapsoVision’ CapsoCam Plus secures FDA nod for paediatric use
CapsoCam Plus comes with four cameras, offering a 360° panoramic view and high-resolution images for enhanced detection of mucosal abnormalities and potential lesions
Saluda Medical announces closing of $100m financing
Net proceeds from the financing will be used primarily to advance the commercialisation of the Evoke System, the first commercial application of the Company’s proprietary closed-loop, dose-control neuromodulation platform
Revvity announces FDA clearance for first automated free testosterone test
This innovative test is the first of its kind to receive FDA clearance for direct quantitative measurement of free testosterone levels, marking a significant advancement in diagnostic capabilities for androgen disorders
Kaneka acquires EndoStream Medical to develop cerebrovascular treatment devices
The acquisition combines the manufacturing expertise and technologies of both firms to develop new cerebrovascular devices in addition to the Nautilus aneurysm device
FDA clears QIAGEN’s QIAstat-Dx Gastrointestinal Panel tests
The QIAstat-Dx Gastrointestinal Panel 2 Mini B&V quickly detects key pathogens using real-time PCR, delivering results in about one hour with minimal hands-on time